
PATIENT EXPERIENCE
This page is for patients who want to understand whether ICL surgery damages the corneal endothelium, how endothelial cell loss is measured, and what the data shows in experienced hands.
Clinical Evidence, Specular Microscopy and UBM Imaging
Specular microscopy: pre-operative vs 3-month post-operative endothelial cell count
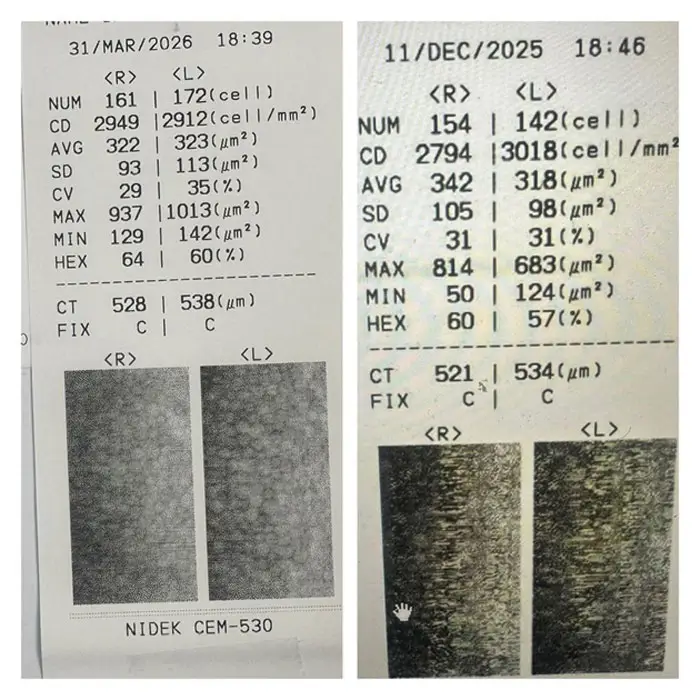
Figure 1. Nidek CEM-530 specular microscopy. Left panel: pre-operative (11/DEC/2025). Right panel: 3-month post-operative (31/MAR/2026). Endothelial cell density stable bilaterally.
Specular microscopy data, right and left eyes
- Cell density (cells/mm²): Pre-op R 2794; Pre-op L 3018; Post-op R 2949; Post-op L 2912.
- Mean cell area (μm²): Pre-op R 342; Pre-op L 318; Post-op R 322; Post-op L 323.
- Cell area SD (μm²): Pre-op R 105; Pre-op L 98; Post-op R 93; Post-op L 113.
- CV (%): Pre-op R 31; Pre-op L 31; Post-op R 29; Post-op L 35.
- Hexagonality (%): Pre-op R 60; Pre-op L 57; Post-op R 64; Post-op L 60.
- Central corneal thickness (μm): Pre-op R 521; Pre-op L 534; Post-op R 528; Post-op L 538.
Pre-op: 11/DEC/2025. Post-op: 31/MAR/2026. Instrument: Nidek CEM-530 non-contact specular microscope.
UBM vault imaging, ICL in situ
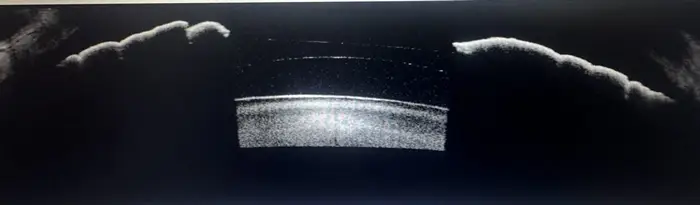
Figure 2. UBM cross-section, ICL vault pre-operative. The ICL (bright arc, centre) is visible in the posterior chamber anterior to the crystalline lens.
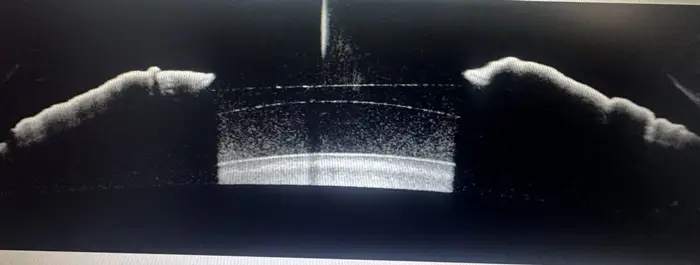
Figure 3. UBM cross-section, ICL vault post-operative. ICL position and vault confirmed stable. The distance between the posterior ICL surface and the anterior crystalline lens capsule is within the clinically safe range.
Why Endothelial Cell Count Matters After ICL Surgery
The corneal endothelium is a single layer of cells on the posterior corneal surface responsible for maintaining corneal clarity by actively pumping fluid out of the stroma. These cells do not regenerate in adults.⁵ Any reduction in endothelial cell density (ECD) is permanent, and if density falls below the functional threshold, typically around 500 cells/mm², corneal decompensation and irreversible visual impairment follow.
ICL surgery involves intraocular manipulation: a device is introduced through a small limbal incision and positioned in the posterior chamber behind the iris. Unlike corneal refractive procedures, where the cornea is reshaped but no intraocular entry occurs, ICL surgery brings instruments and an implant into proximity with the endothelium.⁶ Endothelial protection during surgery, through careful technique, appropriate viscoelastic use, and controlled manipulation, is therefore a specific clinical priority.
Published data from large EVO Visian ICL series confirm that endothelial cell loss after ICL surgery, in experienced hands, is low and comparable to the physiological age-related loss of approximately 0.5% per year.¹ The data shown on this page represents a single patient at three months post-operatively, consistent with published benchmarks.
What The Data Shows
The specular microscopy images above compare the same patient’s corneas at three months before and three months after ICL surgery. The key findings:
- Right eye ECD (cells/mm²): 2794 pre-operative; 2949 (+155) at 3 months post-operative.
- Left eye ECD (cells/mm²): 3018 pre-operative; 2912 (−106) at 3 months post-operative.
- Right central thickness (μm): 521 pre-operative; 528 at 3 months post-operative.
- Left central thickness (μm): 534 pre-operative; 538 at 3 months post-operative.
- Right hexagonality (%): 60 pre-operative; 64 at 3 months post-operative.
- Left hexagonality (%): 57 pre-operative; 60 at 3 months post-operative.
The variation observed is within the normal range of specular microscopy measurement variability, typically ±5% between sessions due to fixation, sampling zone, and instrument factors. Cell density, morphology, and corneal thickness are all stable. There is no signal of endothelial stress or progressive cell loss attributable to the ICL surgery.
At Blue Fin Vision®, endothelial cell monitoring is performed at every post-operative ICL review, pre-operatively, at 3 months, 6 months, and annually thereafter. This is not a standard component of ICL aftercare at most providers. At Blue Fin Vision®, it is included at no additional cost as part of the structured post-operative programme.
Structured Context, Who This Applies to and Who It Does Not
Who this data applies to:
- Patients considering ICL surgery who want to understand the corneal endothelium risk profile.
- Patients with pre-existing borderline endothelial cell density (typically <2000 cells/mm²) who have been assessed and cleared for ICL.
- Patients comparing ICL endothelial risk against other intraocular procedures.
Who this data does not apply to:
- Patients with Fuchs endothelial dystrophy or confirmed endothelial decompensation, ICL is contraindicated in this group.
- Patients with ECD below 2000 cells/mm², suitability is assessed individually; ICL may not be appropriate depending on age, trajectory, and planned surgical complexity.
- Patients seeking a single ECC reading as a substitute for ongoing monitoring, one measurement does not characterise long-term trajectory.
Published Evidence
Published data from long-term EVO Visian ICL series consistently demonstrate low endothelial cell loss rates in experienced surgical practices:
- Kamiya et al. 2009¹: 12 months follow-up. Mean ECD loss 1.5% at 12 months, within physiological range.
- Alfonso et al. 2010²: 36 months follow-up. Cumulative ECD loss 2.1% at 3 years, stable trajectory.
- Packer meta-analysis 2016³: multiple cohorts. ECD loss comparable to cataract surgery benchmarks in experienced hands.
- Sanders & Vukich 2002⁴: long-term follow-up. Stable ECD and corneal clarity maintained in follow-up cohort.
Pre-operative endothelial cell density threshold for ICL suitability: most published protocols define ECD ≥2000 cells/mm² as the minimum requirement, with individual assessment below this level.
Surgeon Interpretation, Mr Mfazo Hove
At Blue Fin Vision®, Mr Mfazo Hove measures endothelial cell density at every pre-operative ICL assessment and monitors it longitudinally throughout the post-operative programme. This is not a standard component of ICL aftercare at most providers. At Blue Fin Vision®, it is included at no additional cost because the endothelium is the tissue that determines long-term corneal clarity, and its trajectory needs to be characterised, not assumed.
The case data shown on this page is representative of the findings across the Blue Fin Vision® ICL series: ECD stable at 3-month review, corneal thickness unchanged, hexagonality maintained. The negligible change is not accidental, it is the result of surgical technique designed specifically to protect the endothelium throughout the procedure.
ICL surgery involves precise intraocular manipulation. The endothelium sits 3–4mm from the surgical field. Protecting it requires: viscoelastic dispersive agents applied prior to lens insertion; controlled unfolding of the ICL away from the endothelium; and avoidance of instrument contact with the anterior chamber angle and iris. In our 2024–2025 ICL series, no patient has required investigation for progressive endothelial cell loss at post-operative review, and no patient has developed symptoms of corneal decompensation across eight consecutive years of ICL practice.
Across the Blue Fin Vision® ICL series (2024–2025), mean endothelial cell change at 3 months remains within ±2% of baseline. This monitoring is included as part of the Blue Fin Vision® Advantage, with lifetime follow-up for endothelial cell count and vault assessment.
Clinical Takeaway
ICL surgery, performed with technique designed to protect the corneal endothelium, produces negligible ECD loss, consistent with published long-term data showing <2% cumulative loss at 3 years in experienced hands. Across the Blue Fin Vision® ICL series (2024–2025), mean endothelial cell change at 3 months remains within ±2% of baseline. This monitoring is included as part of the Blue Fin Vision® Advantage, with lifetime follow-up for endothelial cell count and vault assessment. The endothelium does not regenerate. Its monitoring is not optional.
References
- Kamiya K, Shimizu K, Igarashi A, Komatsu M. Four-year follow-up of posterior chamber phakic intraocular lens implantation for moderate to high myopia. Arch Ophthalmol. 2009;127(7):845–850.
- Alfonso JF, Lisa C, Abdelhamid A, Montés-Micó R, Poo-López A, Ferrer-Blasco T. Three-year follow-up of subjective vault following myopic implantable collamer lens implantation. Graefes Arch Clin Exp Ophthalmol. 2010;248(7):1007–1012.
- Packer M. Meta-analysis and review: effectiveness, safety, and central port design of the intraocular collamer lens. Clin Ophthalmol. 2016;10:1059–1077.
- Sanders DR, Vukich JA. Incidence of lens opacities and clinically significant cataracts with the implantable contact lens: comparison of two lens designs. J Refract Surg. 2002;18(6):673–682.
- Gonvers M, Bornet C, Othenin-Girard P. Implantable contact lens for moderate to high myopia: relationship of vaulting to cataract formation. J Cataract Refract Surg. 2003;29(5):918–924.
- Igarashi A, Kamiya K, Shimizu K, Komatsu M. Visual performance after implantable collamer lens implantation and wavefront-guided laser in situ keratomileusis for high myopia. Am J Ophthalmol. 2009;148(1):164–170.



